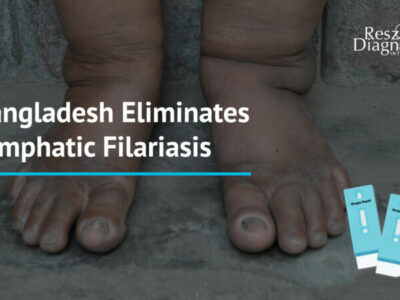
The Global Program to Eliminate Lymphatic Filariasis (GPELF) was initiated by WHO since year 2000 with the ultimate objectives to stop the spread of filariasis infection and to control morbidity through continuous collaborative effort worldwide. This year, Reszon Diagnostics (Reszon) is honoured to announce that our filariasis diagnostic products, PanLF Rapid Test and Brugia Rapid Test, are selected once again for use during transmission assessment survey (TAS) in several endemic countries such as Malaysia, Indonesia, Philippines, Vietnam and Sri Lanka. Through the use of our products as the diagnostic tools, we hope that the correct intervention can be administered and hence helping to stop the spread of filariasis infection in those countries.
Below are some publications that used Reszon’s Brugia Rapid and PanLF Rapid Test:
- Chandrasena, N. T., Premaratna, R., Samarasekera, D. S., & Silva, N. R. (2016). Surveillance for transmission of lymphatic filariasis in Colombo and Gampaha districts of Sri Lanka following mass drug administration. Transactions of The Royal Society of Tropical Medicine and Hygiene. 110(10), Pp. 620-622. doi:10.1093/trstmh/trw067
- Dewi, R. M., Tuti, S., Ganefa, S., Anwar, C., Larasati, R., Ariyanti, E., Herjati, H., Brady, M. (2015) Brugia Rapid™ antibody responses in communities of Indonesia in relation to the results of ‘transmission assessment surveys’ (TAS) for the lymphatic filariasis elimination program. Parasites & Vectors. 8(499). doi:10.1186/s13071-015-1093-x
- Chu, B. K., Deming, M., Biritwum, N., Bougma, W. R., Dorkenoo, A. M., El-Setouhy, M., Fischer, P. U., Gass, K., et. al (2013). Transmission Assessment Surveys (TAS) to Define Endpoints for Lymphatic Filariasis Mass Drug Administration: A Multicenter Evaluation. PLoS Neglected Tropical Diseases. 7(12). doi:10.1371/journal.pntd.0002584